Tastes Like Teen Spirit

Two articles on antidepressants:
one on the modulation of taste by serotoninergic and noradrenergic agents,
and the other on an increased risk of suicide in young people on antidepressants, but not in older people.
The first item is a study published in the Journal of Neuroscience and carried in news outlets as a possible way to predict antidepressant efficacy:
A Taste Sensitivity Test Could Determine the Appropriate Antidepressant[I can imagine certain bloggers are cringing at this notion.]
When faced with a depressed patient, clinicians often have to choose a proper course of treatment based on a guess as to which neurotransmitter in the brain is being disrupted--serotonin, noradrenaline or both. According to Jan Melichar, a psychiatrist at the University of Bristol, doctors "get it right about 60 to 80 percent of the time," but they have to wait up to one month to see if they chose correctly. A report appearing in the December 6 issue of The Journal of Neuroscience could result in an easier way of putting patients on a path to mental stability.
Or, even better:
There Is Accounting for Taste in Treatment of DepressionWhat did the article by Heath et al. (2006) actually show? Well, 2 hours after non-depressed people were given paroxetine (Paxil), an SSRI (selective serotonin reuptake inhibitor), or reboxetine (not approved for use in the US or Canada), a NARI (noradrenegic reuptake inhibitor), they were given a taste test. More specifically,
By Neil Osterweil, Senior Associate Editor, MedPage Today
December 06, 2006
BRISTOL, England, Dec. 6 -- For newly diagnosed depression patients, there may be a tell-tale taste test for selecting the right antidepressant.
On the basis of their finding that serotonin and norepinephrine can alter taste thresholds, investigators here think they may have found a tasteful way to choosing the antidepressant that fits the patient.
In a study of 20 healthy adult volunteers, those who were exposed to the selective serotonin reuptake inhibitor Paxil (paroxetine) had significantly lower thresholds for sweet and bitter tastes than they normally did, wrote Lucy F. Donaldson, Ph.D., of the University of Bristol, and colleagues, in the Dec. 6 issue of Journal of Neuroscience.
Subjects were informed of which taste modality (sweet, bitter, salt, or sour) they were receiving but were given no indication of whether the tester thought they would be able to recognize the taste or not. Low concentrations of one tastant may be misidentified as another; to avoid this potential confounding effect, subjects were informed as to the taste stimulus. ...Tastants were applied in solution to the tip of the tongue, using a cotton bud saturated with the solution at room temperature, and placed on the tongue for ~5 sec. Without closing their mouths, the subjects were asked to indicate whether or not they could taste the stimulus at that concentration.The 20 participants visited the lab 3 times and were tested with varying concentrations of sucrose, quinine, NaCl, and HCl before and then 2 hrs after receiving either paroxetine, reboxetine, or placebo. Ultimately, what happened is that the SSRI increased sensitivity to both sweet and bitter tastes, while the NARI increased sensitivity to bitter and sour (see details below).
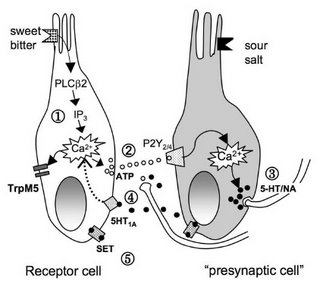
Heath TP, Melichar JK, Nutt DJ, Donaldson LF (2006). Human taste thresholds are modulated by serotonin and noradrenaline. J. Neurosci. 26: 12664-12671.Thus, the taste system turned out to be more plastic than expected. The authors suggest that 5-HT and NA levels play an important role in setting taste thresholds. They present an elaborate model (with cool cartoon taste receptors, below) of how these effects might be mediated at a cellular level (in the taste buds, not in the brain).
Circumstances in which serotonin (5-HT) and noradrenaline (NA) are altered, such as in anxiety or depression, are associated with taste disturbances, indicating the importance of these transmitters in the determination of taste thresholds in health and disease. In this study, we show for the first time that human taste thresholds are plastic and are lowered by modulation of systemic monoamines. Measurement of taste function in healthy humans before and after a 5-HT reuptake inhibitor, NA reuptake inhibitor, or placebo showed that enhancing 5-HT significantly reduced the sucrose taste threshold by 27% and the quinine taste threshold by 53%. In contrast, enhancing NA significantly reduced bitter taste threshold by 39% and sour threshold by 22%. ...
But antidepressants typically take 4-6 weeks to work, not 2 hrs. And what does all this have to do with drug screening in depressed people, anyway? That's the more controversial part, since at the outset they mention the "monoamine theory of depression" which we all know to be a simplistic idea [but see the recent article by Meyer et al. 2006, which explains everything in terms of elevated levels of MAO-A (monoamine oxidase A), a catabolic enzyme.]
Nonetheless, Heath et al. bravely contend (in the press materials) that their results do have direct relevance in developing a "taste taste" for prescribing the correct medications. In the journal article, however, they only acknowledge that taste disturbances are often noted in depression.
The second news item is based on documents released by the FDA:
Antidepressants boost young adults' suicidal risksI have nothing funny to say about that.
WASHINGTON (AP) -- Using antidepressants increases the risk of suicidal thoughts and behavior among young adults but lessens it for seniors, the Food and Drug Administration says.
The effects of antidepressants on adults from 25 to 64 were mixed, so much so that the FDA would only conclude the drug had a neutral effect on suicidal behavior for them but possibly lowers the risk of suicidal thoughts.
The information came from a mass review of 372 studies involving roughly 100,000 patients and 11 drugs, including Lexapro, Zoloft, Prozac and Paxil. The FDA analysis will be incorporated in future changes to antidepressant labels, but the agency wants to first discuss its plans with outside advisers.
In 2004, the FDA ordered strong warnings about the pediatric risk of suicidal tendencies put on antidepressant labels, and began analyzing whether adults face a similar risk, in part by requesting data from drug companies.
It now appears there is an increased risk among adults between the ages of 18 and 25.
. . .
Subscribe to Post Comments [Atom]












0 Comments:
Post a Comment
<< Home